Is Matter Around Us Pure Notes:
Do you know you can save a lot of time by reading class 9 notes of science? The fully packed study material, as well as revision notes, are included in “is matter around us pure notes”. It includes pure and impure substances, mixture with its types, solutions, suspension, colloids and depuration of mixtures. Read and learn it to score better in the exams.
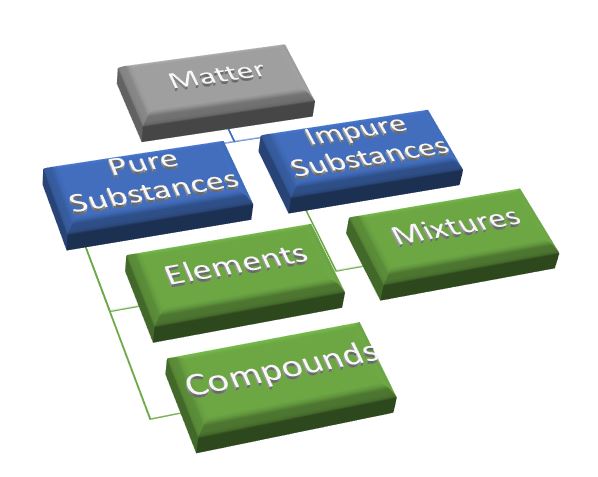
Matter:
Anything that occupies space and has mass and volume is matter.
There are mainly two types of matter:
- Pure substances
- Impure substances or Mixtures
Pure substances:
The material which is consisted of only one type of atoms or molecules throughout is said to be pure substances. Here in 'is matter around us pure notes' we will read more about pure substances.
Pure substances are of two types:
Elements:
- Elements are made up of only single type of atoms.
- Each atom in the element is identical.
- It is a basic form of matter that cannot be broken down into simpler substances by chemical reactions.
- Different elements have different type of atoms.
- Gold, silver, Iron are some examples.
Compounds:
- Compounds are made up of only single type of molecules.
- When two or more elements combine in a fixed ratio by mass then a compound is formed.
- H2O (water), NO2(Nitrogen dioxide) are its example.
- There is little to read about compounds in is matter around us pure notes.
Properties of compounds:
- Compound is considered as the homogeneous substance with definite composition.
- It has fixed melting and boiling point.
- During the formation of compound energy is evolved in the form of heat or light.
- By applying suitable chemical methods we can split compounds into its constituent elements.
Impure substances (Mixtures):
When two or more different types of atoms and molecules which do not react with each other are mixed randomly (any proportion) then the material formed is said to be a mixture. there are categories of impure substances mentioned in is matter around us pure notes.
Properties of mixtures:
- It can be Homogenous as well as Heterogeneous.
- There is variable composition of a mixture.
- Melting and boiling point of a mixture is not fixed in a mixture.
- During the formation of mixture, there is neither release nor absorption of energy.
- The property of mixture is defined by the properties of its constituents.
Mixture is of two types:
- Homogeneous Mixture
- Heterogeneous mixture
Homogeneous Mixture
- It has fixed (uniform) composition.
- There are no visible boundaries of separation between the mixed components.
- Mixture of salt and water is the example of homogenous mixture.
Heterogeneous Mixture:
- It’s composition is not uniform they are not uniformly mixed.
- There are visible boundaries of separation between their components.
- Mixture of oil and water or sand and water is the example of heterogeneous mixture.
- Is matter around us pure notes contains all the information you should know.
Solutions:
- The homogeneous mixture of two or more substances is said to be solution.
- Lemonade, soda water is the examples of solution.
Properties of solutions:
- Solution is homogeneous mixture.
- Solution has particle size (diameter) less than 1nm (10-9m).
- We can’t see particles of a solution even with a simple microscope.
- Scattering the light (Tyndall effect) is not observed in a true solution.
- The best thing about solution is that it is stablee. when left undisturbed, solute particles doesn’t settle down.
- By simple filtration, we can’t separate solute particles from solution.
- What information is essential for the notes is given here in is matter around us pure notes.
Components of solution:
There are two components of a solution:
- Solute: The component of solution which is dissolved and is present in less amount as compared to the solvent is said to be solute. Sugar, salt etc. are solute.
- Solvent: That component of the solution which dissolves the solute in it and is present in a large amount is said to be solvent. Water (universal solvent), alcohol etc. are examples of solvent.
We will read more about solvent in higher classes however is matter around us pure notes is sufficient for now.
Types of solution:
|
Type |
Examples |
| Solid in liquid |
Salt in Water |
| Liquid in liquid |
Alcohol in water |
| Gas in liquid |
Compressed CO2 in
carbonated drinks |
| Solid in Solid |
Different metal alloys |
| Gas in Gas |
Environmental air |
| Liquid in Solid |
Amalgams |
| Gas in solids |
Adsorbed gases on the
surface of different metals. |
| Solid in gas |
Camphor in air |
| Liquid in gas |
Clouds and fogs |